Cryogenic Refrigeration Part III – Cooling with J-T expansion
In order to cool below the practical limit of vapour compression cycles, advantage can be taken of the Joule-Thomson (Joule-Kelvin) cooling effect, a phenomenon that all gases exhibit at a sufficiently low temperature.
To cool a gas from ambient temperature down to its ‘dewpoint’ its enthalpy must be reduced significantly. The dew point is the temperature at which the first drop of liquid appears. Then, to convert the so-called ‘saturated’ vapour to a liquid requires a further removal of enthalpy, the latent (hidden) heat. This term latent is because the heat is removed often without significant temperature change.
But this needs an important qualification.
For pure substances the temperature is indeed constant during liquefaction - so ‘latent’ is a valid term. However, for a mixture such as air, the cooling from saturated vapour at its dew point of ca 86 K at.6 bar to saturated liquid at its bubble point involves a few degrees further cooling typically 2-3 degrees Kelvin.
This may not seem much but where heat exchangers are designed to operate with temperature differences as small as 1 deg K (or C) it must be considered.
For mixtures of fluids which have significantly different condensing temperatures such as methane (112K) and nitrogen (77K) the temperature “glide” during condensation is more significant. For mixtures involving gases such as helium or hydrogen the temperature range during condensation can be large and often it may not be possible to fully condense without freezing the other component(s). For such gases, purification to a high purity is necessary before liquefaction.
Joule-Thomson Effect.
Returning to the issues of refrigeration and enthalpy, most gases at high pressure have a lower enthalpy content (heat content) than the same gas at low pressure and the same temperature.
If the heat exchanger ‘warm end temperature difference’ is small, this allows a returning product stream to effectively take out or remove heat from the process envelope as it carries a greater specific enthalpy (heat content per unit quantity of gas), even though it must be slightly colder than the feed gas at the ‘warm end’ of the process for the heat exchanger cooling the feed to be able to work. ( refer to the simple version of the 2nd Law of thermodynamics, that heat must flow down the temperature gradient).
This gas behaviour is related closely to the so-called Joule-Thomson effect (J-T) where expanding a high-pressure gas across a valve or restriction to a lower pressure causes its temperature to fall even though its total enthalpy remains constant. J -T refrigeration is a simple and frequently used means to keep a continuous cryogenic process cold.
Real and Ideal Gases.
It should be noted that the J-T effect only occurs for non-ideal gases, whereas for small light molecules such as helium or hydrogen, which approach ideal gas behaviour at ambient temperature the J-T effect does not occur. However, it does occur for all gases as the temperature approaches the ‘2-phase boundary’ or dewpoint. But this is no help in providing net refrigeration for a feed and products which behave as ideal gases and enter and leave the process envelope at near ambient temperature. As a result, hydrogen and helium liquefaction need special treatment.
For all gases at very high pressure, above what is called their J-T Inversion curve an isenthalpic expansion causes a temperature rise. This temperature rise can be sufficient to be a safety concern when releasing high pressure hydrogen gas into a recipient container at lower pressure.
The inversion curve is a locus of points for example on a T-S diagram where at lower pressures, the J-T curve is positive (i.e. there is cooling on isenthalpic expansion) and on the other side at higher pressures the J-T effect is negative, i.e. on expansion the temperature rises.
Historically isenthalpic (constant enthalpy) expansion was used for liquefying the permanent gases in conjunction with pre-cooling step(s) using mechanical refrigeration to bring the gas to conditions where there is a significant J-T effect.
It is worth noting that on expansion of a liquid that is significantly subcooled due to pressure, on pressure reduction and before reaching the saturated liquid phase boundary there is a small but possibly significant rise in temperature.
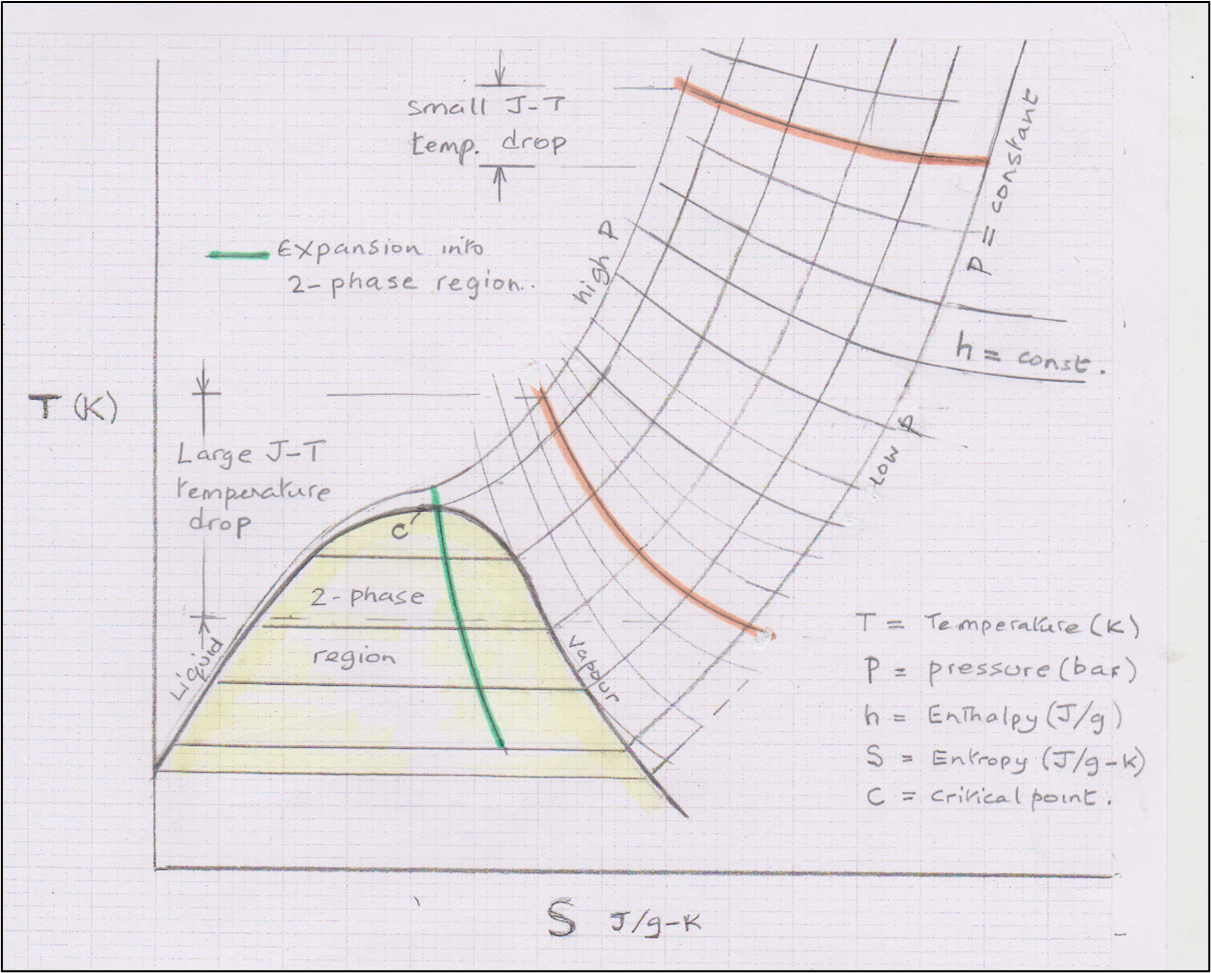
J-T expansion on a Temperature Entropy (T-S) chart
The T-S chart below shows that pre-cooling may be sufficient that, on isenthalpic J-T expansion, the fluid enters the 2-phase region – see green path. This generic illustration shows about 1/3 liquefied and the other 2/3 cools to the same temperature but remains as gas. The cold un-liquefied portion of gas after expansion is typically rewarmed in heat exchange with the incoming HP feed to give the maximum cooling on expansion.
Entropy and the T-S chart are explained in more detail in the next section.
J-T Expansion on T-S chart at high and low initial temperatures
Historically at the birth of cryogenic technology well over 100 years ago, to liquefy ‘permanent gases’, a cascade of successively lower temperature mechanical refrigeration steps were often used before the final J-T step.
Each low temperature stage being condensed by evaporating liquid from the previous stage. Examples include CO2 or ammonia down to minus 30 Celsius followed by ethylene down to minus 104 Celsius and methane down to minus 161 Celsius. This stepwise approach cools the temperature of the fluid to be liquefied to a point where it exhibits a positive Joule-Thompson coefficient. This so-called cascade refrigeration method was used by Klimenko in USSR to liquefy natural gas to make LNG and although potentially quite efficient is rather complex needing three separate compression systems.
Cascade refrigeration for LNG production has now been largely replaced by the use of a single carefully optimised mixture of light hydrocarbons and nitrogen. Propane vapour compression refrigeration in 3 stages at successively lower pressures is commonly used to precool and partly condense the natural gas feed and the mixed or multicomponent refrigerant.